Industry Insight
Next generation treatments and clinical trials for non-small cell lung cancer
Professor Tobias Arkenau from Ellipses Pharma explores the need and potential for next generation treatments as well as the global need to keep improving the clinical trial structure and landscape
It is well known that progress in the discovery of new and effective cancer treatments is hard won and easily lost. The path to success is so often strewn with obstacles – many of them beyond our control.
One thing that is universally agreed upon is that the drug development journey from laboratory to patient should be as fast as possible. The demand for better treatments, faster continually intensifies and it’s up to the oncology community to rise to the challenge.
Therefore, it is understandably exhilarating when key milestones along the development road are passed. One of these is the granting of Orphan Drug Designation (ODD) status, which the US Food and Drug Administration (FDA) is authorised to grant.
The FDA grants ODD drugs for rare diseases that meet certain criteria, such as rearranged during transfection (RET) fusion-positive solid tumours. ODD qualifies the developer for certain incentives with the goal of accelerating drug development for patients, including tax credits and the potential for up to seven years of market exclusivity in the US after approvalbytheFDA. Peoplelivingwithrarecancersor molecularly defined cancer subtypes are increasingly benefiting from new therapies thanks to the ODD programme.
It is known only too well that there is still some way to travel but such a designation does enable drug developers to move forward with greater confidence.
Since the discovery of the RET gene in the early 1980s, multiple treatments have been developed that can inhibit abnormal RET signalling. The first treatments were repurposed multikinase inhibitors, however, their low selectivity for RET led to unacceptable off-target toxicities and suboptimal exposure in patients.
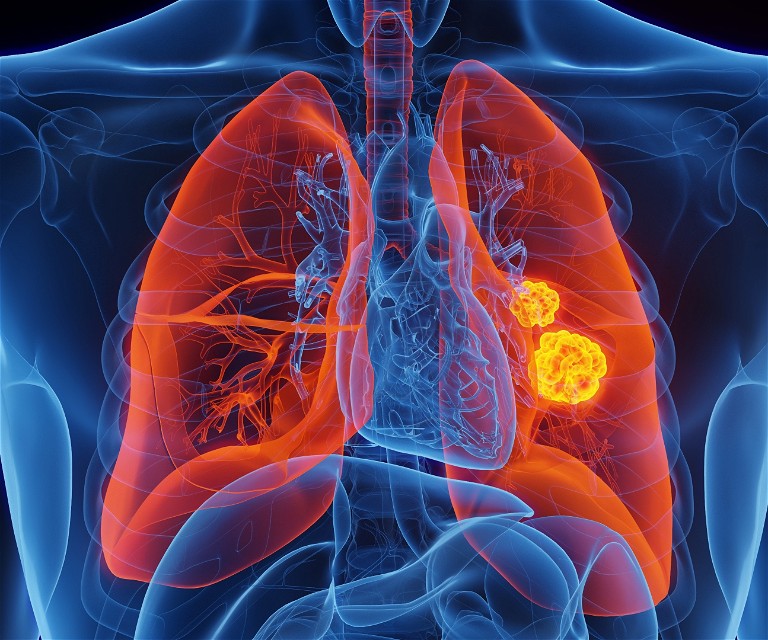
Activating RET mutations and rearrangements have been identified as actionable drivers of oncogenesis in numerous tumour types and are most prevalent in non-small cell lung (NSCLC) and thyroid cancer. It is estimated that RET mutations and rearrangements may be responsible for ~2% of all solid tumours.
Following the modest success of repurposed RET-active multikinase inhibitors, the first selective RET inhibitors received regulatory approval in 2020 and are now standard of care for patients with RET mutant thyroid and RET re-arranged NSCLC and other solid tumours.
Unfortunately, after prolonged exposure to the recently approved selective RET inhibitors (SRIs), a number of acquired escape mutations have been identified together with other non-RET dependent resistance mechanisms. As the understanding of escape mechanisms has increased, it has revealed a clear unmet need for new treatments that can address acquired resistance to these first generation SRIs.
In this context next generation SRIs are in development to overcome acquired escape mechanisms, have better tumour and brain penetration, and importantly improve the safety and tolerability profile relative to first generation SRIs.
There have been various clinical trials that have reported phase 1 data enrolled both treatment naïve and previously treated patients with locally advanced or metastatic RET altered solid tumours, including patients who received prior SRIs. In one study, 109 patients received doses of 10-120mg orally once daily during dose finding and expansion, the majority of whom had advanced RET fusion positive NSCLC. For RET fusion positive NSCLC patients the overall response rate (ORR) was 80.8% [95% CI: 60.7-93.5] and 69.7% [95% CI: 51.3-84.4] in treatment naïve and previously treated patients, respectively. Five out of six patients with baseline RECIST measurable CNS disease achieved an intracranial response.
For the first time some of these drugs have also showed tumour responses in NSCLC patients who were previously treated with first generation SRIs. Responses appeared to be durable regardless of tumour type, RET fusion type, previous treatment or presence of brain metastases.
In addition, a recent phase 1 dose-escalation study focused on patients in the US and Europe and showed similar promising results. Early responses in difficultto-treat NSCLC patients with brain metastases, and responses in patients who received prior SRIs were also observed.
The multi-modular and adaptive trial design in many ways encapsulates the need for innovative and multifaceted international approaches, which yield opportunities to streamline clinical development, in particular, in rare and difficult-to-treat patient populations.
Developing new treatments for patients with rare diseases, including cancer, has many challenges. There are many necessary hurdles that must be overcome along the road to developing a new medicine that can be prescribed, manufactured efficiently and reimbursed by payers – safety, toxicology, efficacy, scaled manufacturing and regulatory to name a few. Overcoming these hurdles is part of ensuring that a drug is both effective, safe to use in its chosen indication, will be prescribed by doctors and affordable to payers. But by and large, the most significant rate-limiting step in the drug development process is the clinical stage.
Patient accrual to clinical trials for patients with rare cancers typically requires a network of dedicated specialist sites, patient advocacy groups and international collaboration in order to achieve the necessary numbers of patients to adequately evaluate a new treatment.
Experience has shown that the elements that drive success in trials – modern, adaptive trial designs – do help make informed decisions fast to benefit patient populations in question. Furthermore, active patient stratification, using targeted medicines, biomarker and omics-driven decision-making and engaging a suitable range of key opinion leaders in the provision and review of real-world evidence to shape a trial in real time are all essential parts of the mix. Combine this with uninterrupted funding, which is crucial to keep trials moving fast from dose escalation to regulatory approval and it is possible to enhance both the speed and the success rates for clinical trials.
Overall, improving the success rate and speed of oncology clinical trials in turn increases the number of treatment options for patients in the future – something that is desperately needed across all cancer types and in all countries. If we can rise to this challenge we will progress more treatments – for patients and oncologists – faster and more frequently.

Author bio:
Professor Tobi Arkenau is responsible for the clinical development of the drugs in Ellipses’ pipeline. He leads clinical strategy, coordinates the drug development teams to deliver the clinical programmes and is an ambassador for the company’s Scientific Affairs Group. Over the course of his career, Tobi has worked on over 300 early-stage clinical trials, with several of these trials providing the compounds that have gone on to become the new standard of care.
A medical oncologist by training, Tobi is a practising clinician having begun his early career as an oncologist in Bremen, Germany and then at the Royal Marsden Hospital in London, UK, before becoming a team leader in Clinical Drug Development at the University of New South Wales, Australia. He has spent over a decade as a Professor at University College London, UK, and most recently was the executive medical director at the Sarah Cannon Research Institute, UK. He holds a PhD from the Medical School of Hanover, Germany.