- All Categories
- UK News
- Europe
- Global
- R & D
- Sales & Marketing
- Manufacturing
- Digital
- Industry Insights
- Appointments
- Interviews
- Features
- Industry Facts
The FDA has announced that it has approved Rezdiffra (resmetirom) for the treatment of adult patients with noncirrhotic non-alcoholic steatohepatitis (NASH) with moderate-to-advanced liver scarring (fibrosis), for use alongside diet and exercise.
Gilead sciences and Merck (known as MSD outside of the US and Canada) have announced results from the phase 2 clinical study assessing the combination of islatravir and lenacapavir for viral suppression in HIV treatment.

Www.pharmatimes.com/croy

Communique.awardsplatform.com

Welcome to the April 2024 issue of Pharmafocus!

Considering the landscape of women’s health in 2024

Pharmarole.com
AstraZeneca has announced that the European Medicines Agency (EMA) has validated two marketing authorisation applications (MAAs) for AstraZeneca and Daiichi Sankyo’s datopotamab deruxtecan (dato-dxd) in the treatment of two cancers.
Spero Therapeutics has announced that it has gained FDA clearance for the investigational new drug (IND) application to assess SPR206 in a phase 2 clinical study.

Amneal Pharmaceuticals has announced that it has entered into an exclusive licensing agreement with Zambon Biotech for IPX204 in the EU, UK and Switzerland.
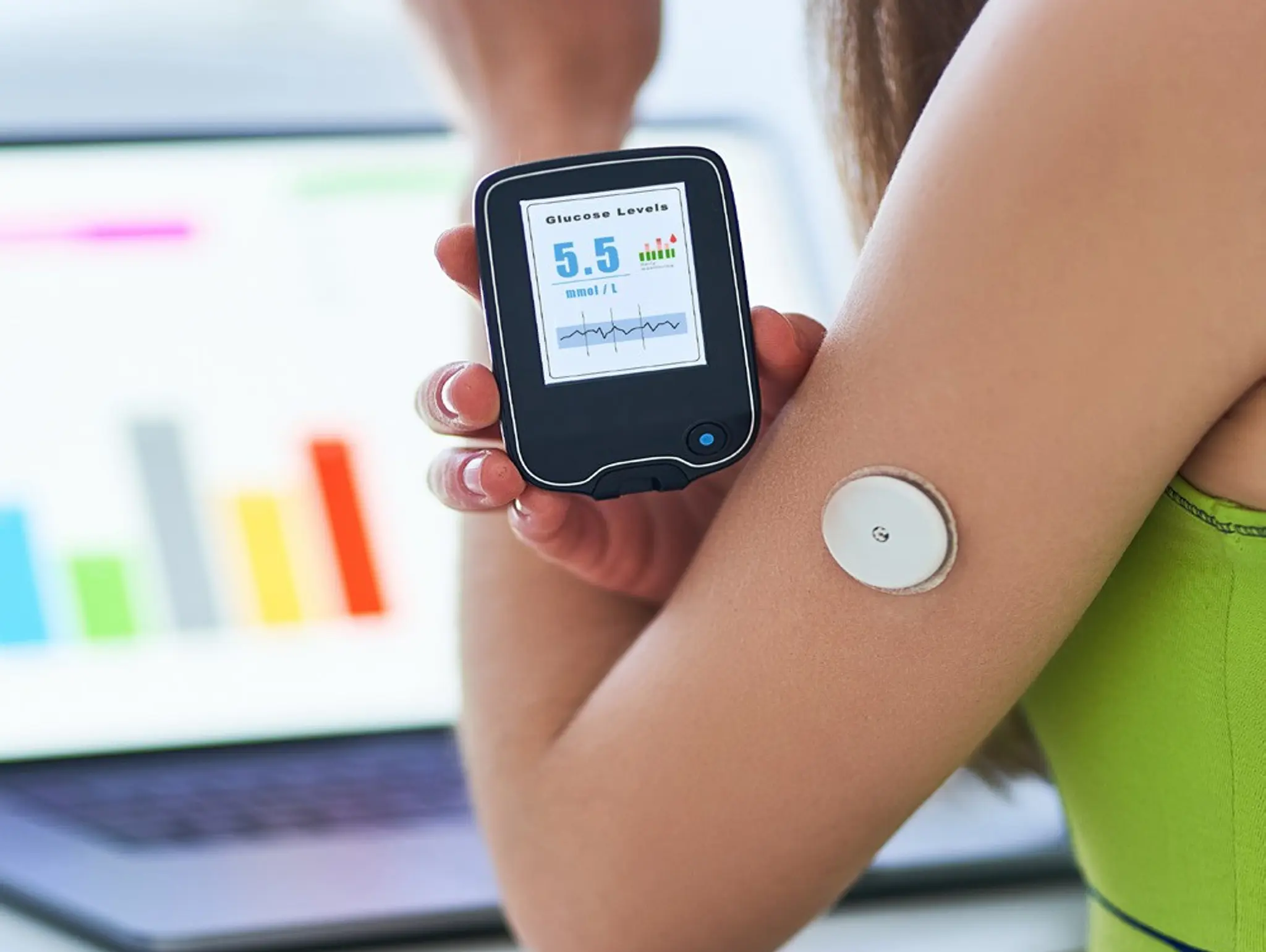
Roche has shared the latest data on its novel solution for continuous glucose monitoring (CGM), including a CGM sensor and two apps designed to present current glucose values and predictions over 30 minutes and two hours.
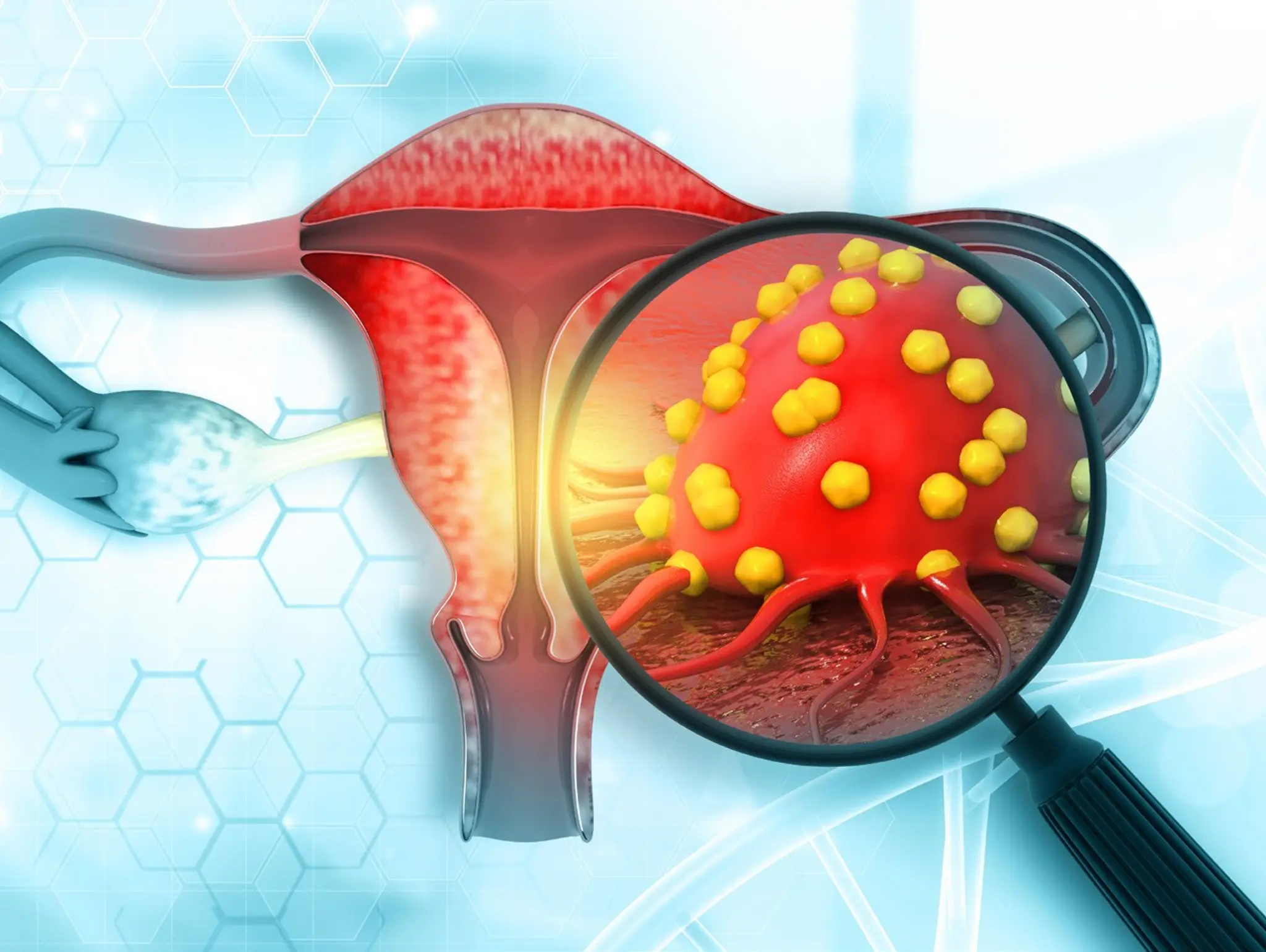
GSK has announced positive results from part 1 and part 2 of its ruby/engot-en6/gog3031/ nsgo phase 3 trial for Jemperli (dostarlimab) in combinations for the treatment of adult patients with primary advanced or recurrent endometrial cancer.