Industry Insight
Bridging science and data: a new era in cancer clinical trials
Dr Gen Li, president at Phesi, considers patient enrolment and retention in clinical trials, with a focus on oncology trials and the challenges facing them
With more than 18 million people in the world affected by cancer each year, oncology has long been a priority area for clinical development organisations.1 New therapies for various types of cancer are needed and currently available treatments – although improving – cannot keep the rising number of cases at bay. In fact, the American Cancer Society predicts that over 600,000 cancer deaths will occur in the US this year alone, with a projected two million new cancer cases.2
Despite a general reduction in mortality when compared to the 1990s, the incidence of six of the top ten cancers continues to increase, with rising rates of breast, pancreas, uterine, prostate, liver and kidney cancers, and HPV-associated mouth cancer.2 It is clear that new cancer therapies are needed, and the clinical development industry continues to invest heavily in this area, with 2,287 oncology products under development in 2022, 2.6 times as many as in 2013.3
This increased investment has led to notable breakthroughs. With immunotherapies such as CAR T and mRNA vaccines, and even stem-cell based therapies against certain types of cancer, oncology trials are frequently at the forefront of scientific discovery.4 And this culture of innovation is starting to have knock-on effects on the oncology clinical trial process. Here, we discuss the steps sponsors are taking to improve cancer oncology clinical development, reduce costs and get effective therapies to patients faster.
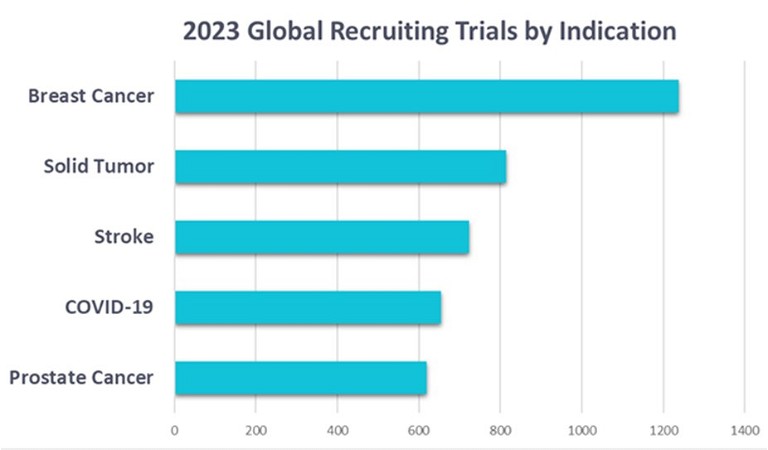
Figure 1: The top five most-studied disease indications in 2023
The current state of cancer clinical trials
Oncology makes up a major proportion of clinical research activity. For the past two years, cancers have made up three of the five most-studied indications. An analysis of 65,749 patient records found that breast cancer was the most studied disease in 2023. Solid tumours was in second place, and prostate cancer in fifth (Figure 1).
This is a similar picture to 2022, but cancers have taken even greater priority as the threat from COVID-19 has diminished (Figure 2). Instead, solid tumour trials – which explore potential drug candidates when a specific cancer type has not yet been identified – have become an area of greater investment in the past year. Increased investment in solid tumour therapies is an exciting move, with these trials likely to be the foundation of innovative cancer therapies. But with innovation comes increased scrutiny from regulators, and like many other trials, the oncology field is plagued by failed, costly and delayed clinical trials.5,6,7
Throughout the clinical development industry, the past two years have seen a surge in trial cancellations at phase 2, with attrition rates sitting at 28% – substantially higher than pre-pandemic levels of 20% (Figure 3). Cancer trials are not immune from these high attrition rates, which slow the rate at which new therapies reach the market and are likely to cause an ongoing increase in costs. Contributing further to rising costs is the increased likelihood that phase 3 cancellations will rise in the coming years as trials that struggled to meet their goals during phase 2 continue to prove untenable during phase 3.
Oncology recruiting challenges
Recruitment is one of the greatest challenges facing clinical trials and is the biggest cause of delays. Approximately four out of five studies fail to meet enrolmenttargetswithinthe intended timeline, and for some clinical development companies, recruitment delays can result in delaying life-saving medicines to reach patients, and loss of revenue.8 In addition to the industry-wide issues of poor site selection, lack of available patients due to competition with other sponsors ‘fishing in the same pond’ and inability to secure a diverse patient cohort, cancer trials have their own unique challenges.
At a treatment level, greater understanding of the genetic drivers of cancer is leading to more targeted therapies. As a result, new cancer therapies may more often be designed for smaller patient subpopulations. With oncology research increasingly focusing on cancers with specific genetic markers and mutations, finding and recruiting the right patients for a trial is a growing challenge – and patients are becoming more dispersed.
Trial site performance in oncology is poor. A 2023 analysis of 173 cancer clinical trials found that across three years, nearly one in five investigator sites recruited just one patient. These findings correlate with research from the Tufts Center for the Study of Drug Development, which found that 11% of sites fail to enrol a single patient and 37% under-enrol.9
In oncology trials, disparity between recruiting sites is vast, with almost half of cancer trial patients coming from just 16% of sites. Meanwhile, at the other end of the spectrum, 20% of trial sites contribute just 3% of patients. The fiscal impact this has on the industry is substantial. A single-patient site has an average cost-per-patient that is ten times higher than a better-performing site ($130,000 per patient vs. $14,167).
A part of the challenge is the inadequate toolkits available to support sponsors in selecting the right investigator for a given trial. For example, of 2,777 recruiting phase 2 cancer trials, 563 (20%) of these placed an investigator site in MD Anderson, one of the largest cancer hospitals in the world. It is impossible to expect MD Anderson to deliver satisfactory enrolment results for 563 trials.
As oncology therapies shift toward being more precision-based, the industry must brace for the practical implications that come with this. Without adequate planning, trials are likely to see a rising number of single-patient sites and the increased operational costs and cycle times that come with this. Once again, this underscores the urgent need for the industry to act now to address these issues.
Knowing your patients
To improve the performance and success of oncology trials, sponsors must adapt and use the data gathered from historical and existing trials and clinical research to move from ‘perception-led’ to ‘data-led’ programme and study design. Trial design should start with the patient at the very centre; analysing data to have a true idea of the target patient enables the clinical development process to be streamlined.
This begins with a Digital Patient Profile (DPP) – a granular breakdown of the patient population for the target indication. With a DPP, sponsors gain a statistical view of patient attributes including typical age, sex, ethnicity and comorbidities, as well as many other key variables that can help to eliminate non-active, non-enrolling sites.
The data held within a DPP has many further applications in making trials more efficient. DPPs can inform predictive analytics for protocol design, trial simulation and scenario modelling to predict trial outcomes and risk factors – including likely side effects. These deep analyses minimise overall operational costs from protocol amendments and enhance submission packages for new drug approvals.
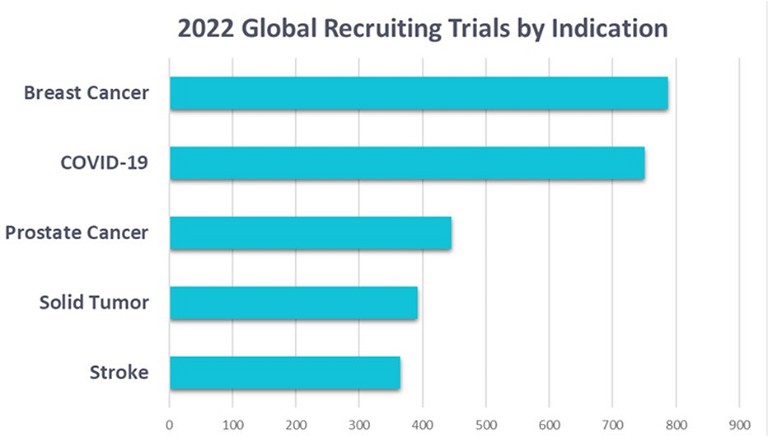
Figure 2: The top five most-studied disease indications of 2022
Data-driven trial design
Where patients are in low supply – as is the case for many rare cancer indications – data can be further used to support single arm clinical trials, eliminating the need for control arms and reducing patient burden. Cancer trials are set to see DPPs being used to inform digital twins and digital trial arms, to allow a clinical study to be modelled from early screening and accelerating the route of treatments to patients.
Oncology drug approvals are increasingly supported by single arm trials. An analysis by JAMA Oncology revealed that between 2002 and 2021, 31% of new oncology drug indications approved by the FDA were based on data from single-arm trials.10 One example is GSK’s single-arm trial assessing dostarlimab for a rare form of locally advanced rectal cancer.11 In this trial, a total of 12 patients completed treatment with a clinical complete response.
But these small, single-arm trials come with a risk. The DPP of the rare rectal cancer targeted in a dostarlimab trial shows that the participants in the trial were not truly representative of the patient population, trending younger and with more women involved than proportionate. This type of miscalculation can be costly, with phase 2 trial results typically used to implement a larger, randomised controlled trial. To find out that the foundational data of a phase 3 trial is inadequate is a high cost that could be avoided with the use of a DPP.
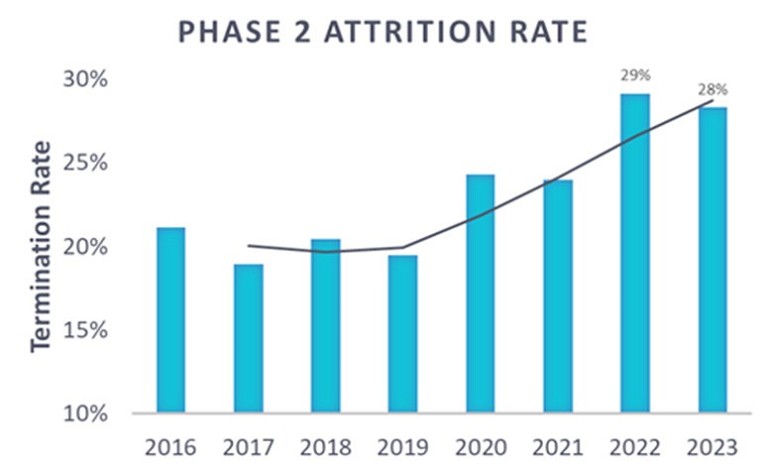
Figure 3: Phase 2 attrition rate (2016-2023)
Clinical trial trailblazer
With their higher levels of investment, oncology clinical trials tend to be at the forefront of innovation when it comes to both the underlying science and the clinical development process. As cancer trials continue to push the boundaries, this is where the industry should look to have a view of the trials of the future, but the oncology space should prepare for the pitfalls ahead and plan trials armed with the best data possible. Innovation does not have to be expensive or high-risk. The data to accelerate clinical trials, reduce costs and minimise patient burden already exists, it just needs to be applied.
References
- Visit: wcrf.org/cancer-trends/worldwidecancer-data/#:~:text=Find%20information%20 about%20world%20cancer,and%208.8%20 million%20in%20women
- Visit: pubmed.ncbi.nlm.nih.gov/38230766/
- Visit: iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/globaloncology-trends-2023
- Visit: ncbi.nlm.nih.gov/pmc/articles/PMC8863908/
- Visit: reuters.com/business/healthcarepharmaceuticals/gilead-says-trodelvy-failsmeet-primary-goal-2nd-line-lung-cancertrial-2024-01-22/
- Visit: theguardian.com/science/2017/apr/28/cancer-drugs-fund-condemned-as-expensiveand-ineffective
- Visit: reuters.com/business/healthcarepharmaceuticals/gsk-says-blood-cancerdrug-meets-primary-goal-late-stagetrial-2023-11-27/
- Visit: ncbi.nlm.nih.gov/pmc/articles/PMC7673977/
- Visit: nature.com/articles/nrd.2018.111
- Visit: cancertherapyadvisor.com/home/cancer-topics/general-oncology/role-single-arm-trials-oncology-drugapprovals/#:~:text=Oncology%20drug%20 approvals%20in%20the,limitations%20that%20 must%20be%20considered.
- Visit: nejm.org/doi/10.1056/NEJMoa2201445?url_ver=Z39.88-2003

Dr Gen Li founded Phesi in 2007 with the aim of revolutionising the clinical trials industry. Prior to this, Gen was head of Productivity for Pfizer Worldwide Clinical Development, a position he assumed following Pfizer’s acquisition of Pharmacia, where Gen delivered the first implementation of productivity measurement for clinical development. While at Pharmacia and Pfizer, Gen significantly contributed to the Centre for Medicines Research (CMR) International database for pharmaceutical R&D performance, assuring the collection of key clinical trial parameters as representative of the critical path for delivery. He was also instrumental in creating the KMR productivity mode.