Future Focus
FDA approves skin cancer AI detection device
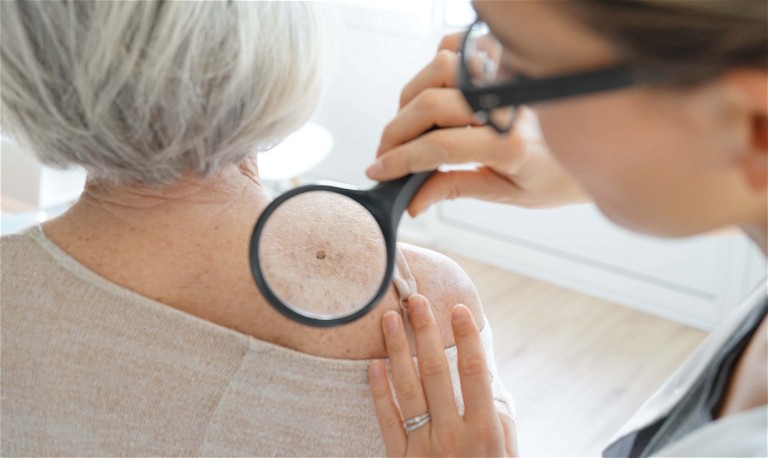
DermaSensor has announced that the US Food and Drug Administration (FDA) has granted clearance for its real-time, non-invasive skin cancer evaluation system. The AI-powered medical device is equipped to detect all three common skin cancers, melanoma, basal cell carcinoma and squamous cell carcinoma.
The FDA approval follows a pivotal study of over 1,000 patients, which demonstrated that the device had a sensitivity of 96% across all 224 skin cancers. A negative result had a 97% chance of being benign.
In another study, the device was demonstrated to reduce the number of missed skin cancers by half (from 18% to 9%).
Cody Simmons, co-founder and chief executive officer of DermaSensor, commented: “We are entering the golden age of predictive and generative artificial intelligence in healthcare, and these capabilities are being paired with novel types of technology, like spectroscopy and genetic sequencing, to optimise disease detection and care.
Equipping PCPs, the most abundant clinicians in the country, to better evaluate the most common cancer in the country has been a major, longstanding unmet need in medicine. While dozens of companies have attempted to address this problem in recent decades, we are honored to be the first device cleared by the FDA that provides PCPs with an automated tool for evaluation of suspicious lesions.”
Dr Maurice Ferre, co-founder and chairman of DermaSensor, added: “Achieving this medical milestone is a testament to the 12 years and tens of millions of dollars our company has invested in research and development to bring this powerful technology to market. We are incredibly grateful to the FDA for their collaboration and dedication to this area starting with our first FDA pre-submission meeting in 2016. Having begun patient enrolment in our FDA pivotal study in mid-2020, we are now ecstatic to have clearance of our FDA-Breakthrough Designated De Novo submission.”