Insider Interview
The evolving landscape of RA treatments
Professor Rieke Alten, Head of the Department of Internal Medicine at Schlosspark-Klinik, illuminates the benefits of subcutaneous infliximab in treating rheumatoid arthritis
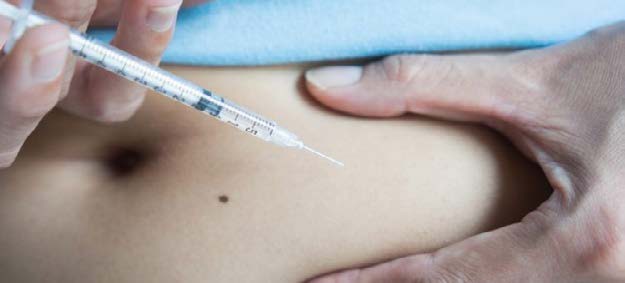
Pharmafocus: What are the benefits of subcutaneous infliximab?
Professor Rieke Alten: Especially under pandemic conditions, it is necessary that physicians adopt a more tailored approach when it comes to deciding whether a patient should receive intravenous (IV) or subcutaneous (SC) infliximab, according to each individual’s condition or disease state. For instance, an IV formulation could be used to induce a rapid response in patients, whilst SC can be utilised to maintain a better drug survival rate.
All patients with immune-mediated diseases have to commit to a treatment for years, often decades. Due to this, patient adherence is a key issue.
Therefore, especially under pandemic conditions, a SC formulation of infliximab offers patients a better quality of life by releasing them from the burden of having to travel to treatment for IV infusions. Self-administration reduces demand on healthcare systems, keeping patients out of clinics and providing clinicians and nurses with additional time.
What challenges do people with rheumatoid arthritis face in their daily lives?
Rheumatoid arthritis (RA) is a chronic, progressive, inflammatory autoimmune disease.
It is characterised by painful, tender, and swollen joints that can significantly affect a patient’s quality of life, productivity, and participation, due to pain, stiffness, and sleep disturbances.
Morning stiffness is one of the most prevalent problems for people with RA, affecting the majority of those experiencing even low disease activity. For people living with RA, fatigue also remains a major constraint, and unmet need.
People with RA can experience higher rates of depression and anxiety, as well as periods of exacerbation of the disease known as flares.
How does subcutaneous infliximab compare to existing treatments for RA?
In clinical studies, Remsima SC showed clinical advantages in terms of pharmacokinetics compared to the IV formulation, implying that it is a biobetter. A biobetter is defined as a ‘modified version of a specific, approved biologic, that enhances clinical outcomes (e.g., improved efficacy) and/or drug pharmacology (e.g., pharmacokinetics and/or pharmacodynamics).
Remsima SC has been shown to have a stable potency, and patients with RA on Remsima SC showed lower rates of ADA, compared with patients receiving Remsima IV. With the availability of both an IV formulation and the novel SC formulation of infliximab, clinicians can choose the optimal treatment for their patients, providing a more personalised and convenient treatment plan.
What are the greatest obstacles in developing treatments for RA?
When developing treatments for RA, it is key to balance safety, efficacy, and convenience for the patient, particularly as life-long treatment is mandatory.
In RA, disparities between high and low GDP countries still play a major role in treatment allocation.
What are your hopes for the future treatment of RA?
Having enough and adequate resources to treat all patients who need an advanced therapy to achieve remission or at least LDA.
Shared decision-making is an integral part of T2T. This should always be an integral part of the art of medicine.
Professor Rieke Alten, Head of the Department of Internal Medicine, Rheumatology, Clinical Immunology and Osteology at Schlosspark-Klinik, Teaching Hospital of Charité, Berlin, Germany