Digital
Latest updates on the digital side of the pharmaceutical industry
Articles
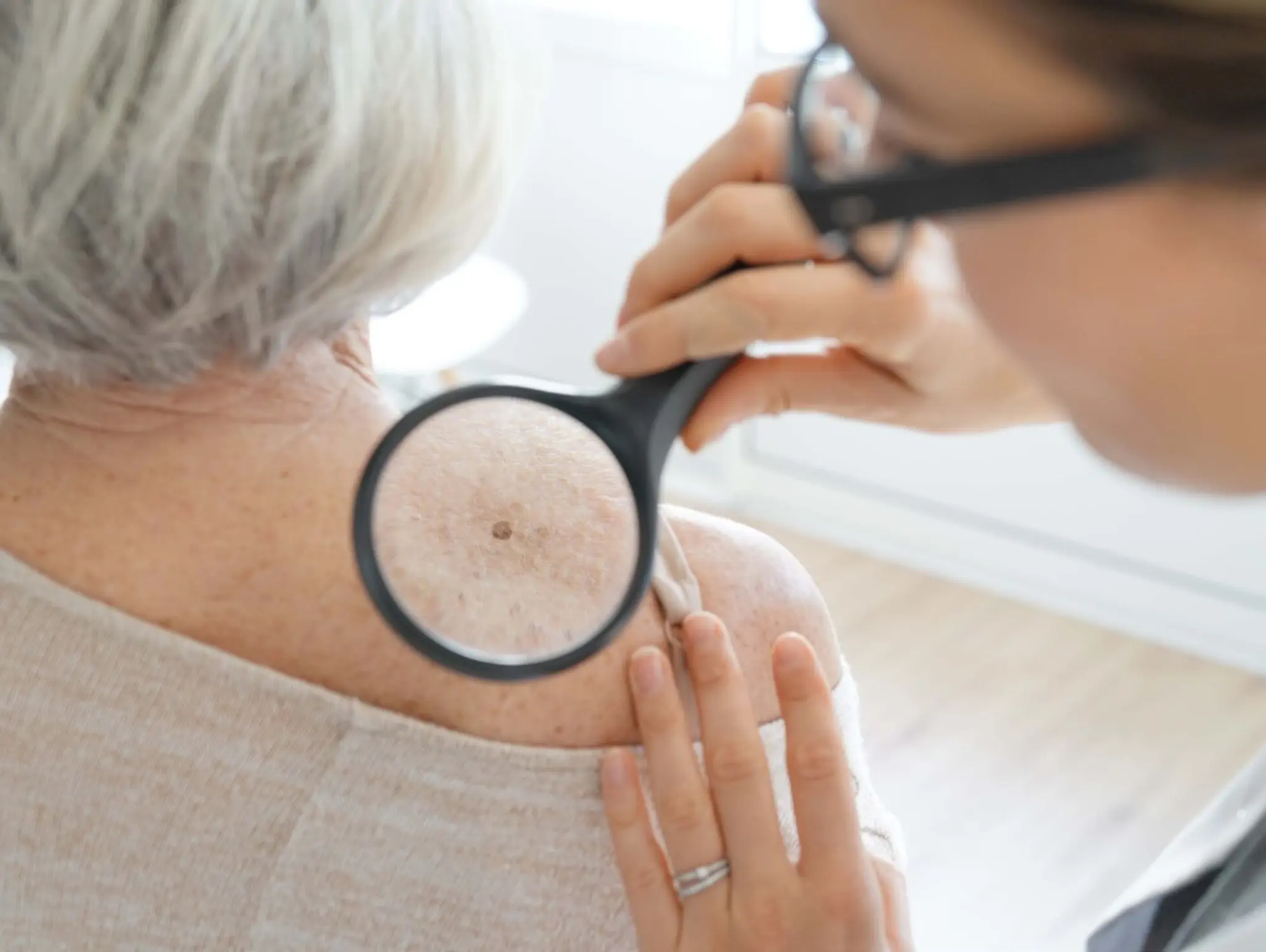
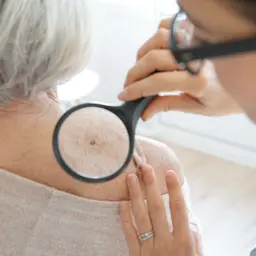
DermaSensor has announced that the FDA has granted clearance for its real-time, non-invasive skin cancer evaluation system.
Eli Lilly has announced its new digital healthcare experience, LillyDirect, which will benefit patients in the US with obesity, migraine and diabetes. This is intended to offer resources around disease management, such as access to healthcare providers, tailored support and delivery of selected medications.

The National Institute for Health and Care Excellence (NICE) has recommended six digitally enabled therapies for adult patients with anxiety disorders and three digitally enabled therapies for adult patients with depression for use within the NHS while further data is gathered.

French pharmaceutical company Servier and US-based causal AI and digital twins company Aitia have announced a collaboration to create digital twins as an aid to help treat pancreatic cancer. This collaboration builds on a previous multiple myeloma one that was announced in 2022.

Bristol Myers Squibb and Viz.ai, an AI-powered medical imaging company, have signed a deal to use AI to detect and triage patients with suspected hypertrophic cardiomyopathy (hcm).

Finnish company Sooma medical has received breakthrough device designation by the FDA for its portable, at-home neuromodulation device to treat depression.

The FDA has reportedly rejected a bid by Elon Musk’s Neuralink in which it proposed the initiation of human testing for its brain chip implants. The company has been working on brain implants that allow for a computer-brain interface, which could allegedly restore vision to the blind and help paralysed people to walk again.

A team of scientists have broken records with their new AI which has found a novel drug candidate for liver cancer in only 30 days

A new AI has been developed to deliver medicines at a faster rate and lower cost

Becton, Dickinson and Company has revealed its new-generation robotic track system

A recently developed AI model has been shown to predict ten-year incidences with a similar level of success to the current standard of care.

Anumana, an AI-driven health technology firm, has signed a multi year deal with Pfizer in order to develop an AI electrocardiogram (AI-ECG) for the early detection of cardiac amyloidosis.